Cell Therapy Products, Workflow & Research Services
Cell therapy workflowTake the next step
Promise and Potential: Cell Therapies for Pediatric PatientsInterview with Professor Dr. Peter Bader, Head of the Division for Stem Cell Transplantation and Immunology, Department for Children and Adolescents, University Hospital Frankfurt, Goethe University Read more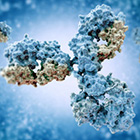
Ready to Get Started?Ready to Maximize Production Efficiency? Our Bioprocessing Solutions team can help you streamline your biomanufacturing processes! Yes, let's get started!What Is Cell Therapy?Cell therapy, or cellular therapy, is the introduction or transplantation of healthy cells into a person's body to replace missing or diseased cells. Cell therapies include several approaches that require manipulating donor tissue, both autologous and allogeneic, to obtain desired cells and tissues. These cells and tissues, or cell therapy products, are used to treat diseases, ranging from wound repair to cancer. Cell therapies emerged in the late 1980s and have grown with the expansion and activation of LAK and TIL adult stem cells (T cells) isolated and expanded from peripheral blood, bone marrow, cord blood, and dermal/keratinocyte grafts to treat burn patients. Today, cell therapy approaches are moving away from stand-alone cell delivery to co-therapies with vaccines or mAbs. Recent developments such as CAR-T and TCR-T offer new, life-saving treatments for patients in the fight against cancer. What Is the Difference Between Allogeneic and Autologous Cell Therapy?The difference between allogeneic and autologous cell therapy is the source of cells. Allogeneic cells come from a person other than the patient receiving the therapy. Autologous cells come from the same person who gets the transplant, while allogeneic transplants generally report a lower risk of disease recurrence than autologous transplants. However, allogeneic transplants may be associated with complications. What Should I Consider When Researching Cell Therapy Products?When you research cell therapy products, it's important to consider current regulations set in place by the Center for Biologics Evaluation and Research (CBER). The CBER must approve cell therapy products, human gene therapy products, and specific devices related to cell and gene therapy. Additionally, as cell therapies advance through clinical trials toward approval, the success of cell therapy depends increasingly on process development strategies that enable improved efficiency and maintain or improve safety and quality. High-quality materials and services are vital for optimal performance throughout every phase of the cell therapy workflow. Discover the Ideal Cell Therapy Products for Your ExperimentsAvantor® offers products to help researchers and manufacturers develop cell therapies. We carry a broad range of products, including:
|
|
Processing your request... |