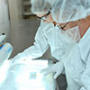
Types of Environmental Control in Biopharma
Research and manufacturing in the biopharmaceutical industry require optimal conditions to achieve the best results. Environmental controls, including temperature, humidity and air quality, are essential to maintaining optimal conditions that protect the integrity of intricate processes. What are the benefits of environmental monitoring?Environmental monitoring in the biopharmaceutical industry helps ensure regulatory compliance and maintains product quality. A proactive approach to monitoring and controlling environmental conditions helps mitigate risks, reduces costs and leads to continuous improvement in manufacturing processes. Monitoring the environment in the biopharmaceutical industry is vital for various reasons, including: Regulatory complianceMonitoring environmental conditions helps biopharma companies adhere to strict regulatory standards, including meeting Good Manufacturing Practices (GMP) and other regulatory requirements. Product quality assuranceBiopharma companies can protect the quality of their research and manufacturing by carefully monitoring and controlling environmental conditions in their research or production facilities. Environmental controls help maintain ideal operating conditions and lower the chances of contamination that can negatively impact product quality. Early contamination detectionDetecting contamination as early and quickly as possible can help reduce its spread and minimize product loss. Early detection also allows biopharma companies to take corrective actions more quickly to resume normal operations and maintain product integrity. Process optimizationData obtained by environmental monitoring can be valuable to biopharma companies that want to optimize their manufacturing processes. Fine-tuning these processes based on environmental conditions can enhance efficiency and reduce the consumption of resources. Risk mitigationSeveral environmental factors could compromise product safety and efficacy. Continuous monitoring and environmental controls can identify and mitigate those risk factors, improving risk management in biopharmaceutical manufacturing. Types of environmental controlEnvironmental factors such as temperature control, humidity, clean room maintenance, air quality control and pressure management can dramatically impact biopharmaceutical research and manufacturing. For example, precise temperature control can safeguard sensitive processes, while humidity management can help prevent moisture-related issues. Clean room control helps maintain low particle levels to help avoid damaging product integrity. Air quality control can minimize the risk of contamination, and pressure control helps prevent cross-contamination. Each factor is critical in creating and sustaining optimal biopharma research and manufacturing conditions. Temperature controlControlling temperature can be very important in manufacturing biopharmaceutical products because of its impact on precision, product quality and process efficiency. Proper temperatures are essential for maintaining the stability of biological materials, such as proteins and enzymes, which are often key components of these pharmaceuticals.
Humidity controlMany biopharma processes are susceptible to environmental conditions, and any deviations in humidity levels can adversely impact pharmaceutical product production and quality. That’s why humidity control is an important aspect of maintaining optimal conditions.
Clean room controlBiopharmaceutical are controlled environments designed to reduce the risk of airborne particles, microbes and other contaminants that could compromise the quality of pharmaceutical products. Factors such as air cleanliness, temperature, humidity and pressure are tightly regulated to adhere to specific cleanliness standards, helping to ensure consistent and reproducible biopharmaceutical production. Air quality controlAirborne contaminants, such as particles, microorganisms and volatile organic compounds, can pose a severe threat to product integrity and the safety of workers and consumers. Stringent air quality control measures, such as air filtration systems, can help ensure the air in a production environment is free of impurities. Pressure controlPressure can also play an important role in biopharmaceutical manufacturing because it can affect reaction rates, solubility and phase transitions, as well as cell viability and the production of bioactive compounds. In addition, pressure control is important in chromatography, filtration and downstream processes, where deviations can compromise product purity and yield. Several systems can be used to maintain the proper pressure differentials needed in biopharmaceutical manufacturing. Pressure sensorsPrecision pressure sensors are essential for real-time monitoring because they continuously measure pressure differentials and provide immediate feedback to control systems. Constant monitoring helps keep the manufacturing environment within specified pressure ranges to maintain product quality and comply with regulatory standards. Control valvesControl valves act as dynamic regulators, allowing engineers to finely adjust pressure differentials in response to changing production conditions. By responding quickly to variations and preventing pressure fluctuations that could compromise biopharmaceutical processes, these valves maintain a stable manufacturing environment. Automated systemsAutomated systems are guided by algorithms and predefined parameters to continuously assess pressure levels and take corrective actions to maintain consistent pressure differentials. These automated systems minimize the risk of human error, and their quick response to pressure variations can enhance the reliability of the manufacturing process. Robust engineering controlsEngineering controls include structural and procedural safeguards against pressure fluctuations. These systems may incorporate built-in redundancy and are designed to identify and address pressure deviations quickly. Fail-safe and backup mechanisms enhance the manufacturing process’s resilience to maintain desired pressure differentials and reduce the risk of disruption. Analyzing the results of environmental control techniquesAssessing the efficacy of environmental control involves constant monitoring and data collection, providing immediate insights for quick corrective actions. Systematic risk assessment and mitigation techniques can also identify and address potential challenges. Monitoring and data collectionAutomated monitoring systems with sensors gather real-time data on factors such as temperature, humidity, pressure differentials and air quality in clean rooms and controlled environments. This continuous data stream helps quickly identify and correct deviations from desired conditions. This proactive approach minimizes the risk of product contamination or any compromise in quality. Data analytics and visualization tools can also provide technicians with the information they need to maintain optimal conditions. Risk assessment and mitigationComprehensive risk assessments are essential to maintain environmental control in biopharmaceutical production and research. These assessments help pinpoint potential challenges that could threaten product quality, safety and regulatory compliance. Various techniques, such as Failure Mode and Effects Analysis (FMEA) or Hazard Analysis and Critical Control Points (HACCP), can systematically identify and evaluate vulnerabilities within manufacturing environments. Quality control and validationQuality control and validation play important roles in maintaining the dependability, accuracy and compliance of environmental control systems in biopharmaceutical production. Outlook for the futureThe biopharmaceutical industry uses many environmental control techniques to ensure the optimal conditions for pharmaceutical production. These techniques involve closely monitoring critical parameters like temperature, humidity and air quality alongside vigorous measures to control contamination. Seamlessly integrating clean rooms, HVAC systems, sterilization protocols and other techniques plays a crucial role in creating ideal manufacturing environments. |
|
Processing your request... |