STERIS CORP
Supplier:
Steris
Description:
Indicator strips are used to validate and re-qualify ethylene oxide gas or dry heat sterilizers and to monitor the effectiveness of common EO gas or d...
Supplier:
Steris
Description:
VERIFY™ Dual Species Self-Contained Biological Indicators (SCBIs) are designed for use in monitoring steam sterilization and ethylene oxide (EO) steri...
Supplier:
Steris
Description:
Chemical integrators monitor all critical parameters of steam sterilization (time, temperature and steam quality).
Supplier:
Steris
Description:
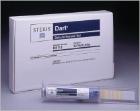
The Dart® daily air removal test is a pre-assembled Bowie-Dick test pack products used in monitoring healthcare steam sterilizers.
Catalog Number:
(89065-290)
Supplier:
Steris
Description:
Test packs consist of preassembled single-use test packs designed to evaluate the performance of the air removal system of a pre-vacuum equipped steri...
Supplier:
Steris
Description:
The Spordex® suspension contains a certified population of bacterial spores suspended in an aqueous solution. The Spordex® suspensions for ethylene ox...
Catalog Number:
(89176-592)
Supplier:
Steris
Description:
Bowie-Dick Tests verify that the prevacuum sterilizer effectively removes air and provides efficient steam penetration.
Catalog Number:
(77463-572)
Supplier:
Steris
Description:
Spordex® VH202 stainless steel biological indicator is used in cycle development and routine monitoring of applications with STERIS VHP biodecontamina...
Inquire for Price
Stock for this item is limited, but may be available in a warehouse close to you. Please make sure that you are logged in to the site so that available stock can be displayed. If the
Stock for this item is limited, but may be available in a warehouse close to you. Please make sure that you are logged in to the site so that available stock can be displayed. If the
To process your orders without delay, please provide the required business documentation to purchase this product.
To order chemicals, medical devices, or other restricted products please provide ID that includes your business name & shipping address via email CMD_NA@vwr.com or fax 484.881.5997 referencing your VWR account number. Acceptable forms of ID are:
* ATTN: California Customers may require additional documentation as part of the CA Health & Safety Code. Products that fall under this regulation will be placed on a mandatory 21-day hold after documentation is received. VWR will not lift restrictions for residential shipping addresses.
-Additional Documentation May be needed to purchase this item. A VWR representative will contact you if needed.
This product has been blocked by your organization. Please contact your purchasing department for more information.
The original product is no longer available. The replacement shown is available.
This product is no longer available. Alternatives may be available by searching with the VWR Catalog Number listed above. If you need further assistance, please call VWR Customer Service at 1-800-932-5000.
|
|||||||||